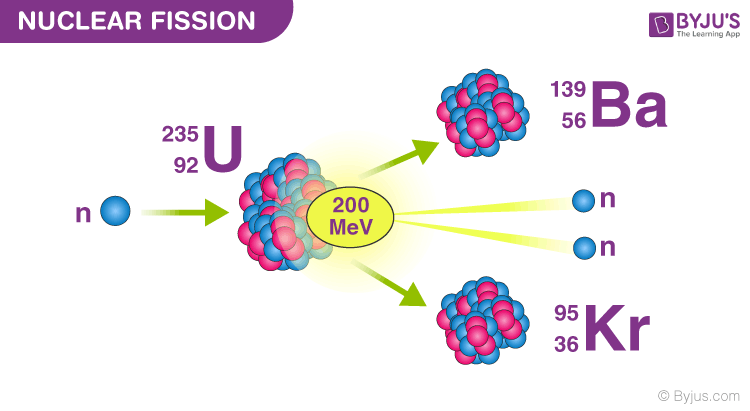 
As can be seen, for low mass numbers E b/A increases rapidly and reaches a maximum of 8.8 MeV at approximately A=60. This calculated fraction is shown in the chart as a function of them mass number A. The nuclear binding energies are enormous, they are on the order of a million times greater than the electron binding energies of atoms.įor a nucleus with A (mass number) nucleons, the binding energy per nucleon E b/A can be calculated. Nuclear binding energy curve.ĭuring the nuclear splitting or nuclear fusion, some of the mass of the nucleus gets converted into huge amounts of energy and thus this mass is removed from the total mass of the original particles, and the mass is missing in the resulting nucleus. According to the Einstein relationship ( E=m.c 2) this binding energy is proportional to this mass difference and it is known as the mass defect. The difference is a measure of the nuclear binding energy (E b) which holds the nucleus together. The mass of a nucleus is always less than the sum of masses of the constituent protons and neutrons when separated. The protons and neutrons in an atomic nucleus are held together by the nuclear forces (strong force).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

:max_bytes(150000):strip_icc()/fission-of-a-uranium-nucleus-141483757-579266b03df78c173498d794.jpg)
 RSS Feed
RSS Feed
